of patients on EGFR inhibitors develop a papulopustular rash.
Lacouture ME et al. Oncologist 2008; Support Care Cancer 2011.
Oncology dermatology · CTCAE v5.0
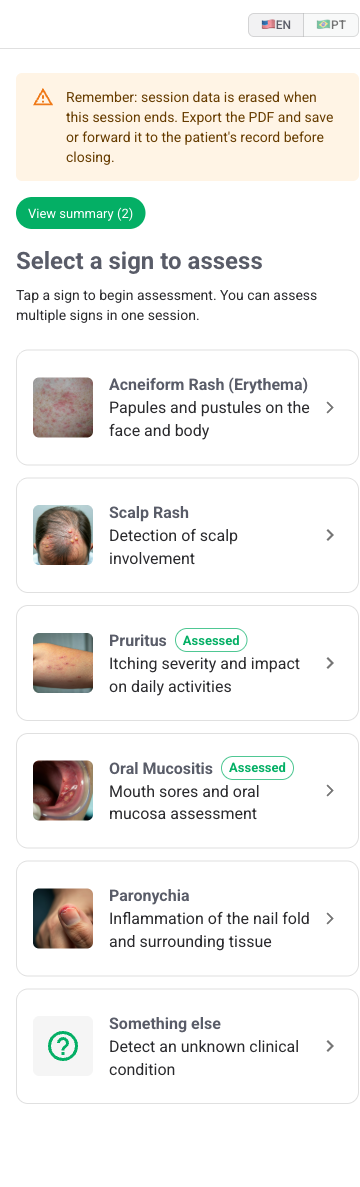
Oncoderm is a focused, mobile-first tool for oncology teams to assess dermatologic toxicities, including erythema, scalp rash, pruritus, oral mucositis, and paronychia, and produce CTCAE-aligned grades in under a minute.
Built on the standards your team already uses
Why oncoderm exists
of patients on EGFR inhibitors develop a papulopustular rash.
Lacouture ME et al. Oncologist 2008; Support Care Cancer 2011.
of patients on immune-checkpoint inhibitors report itch or rash within 6 weeks.
Sibaud V. Am J Clin Dermatol 2018.
inter-rater agreement on CTCAE skin grading between oncologists; fair, not strong.
Atkinson TM et al. JNCI 2016.
of grade-3 dermatologic AEs trigger dose interruption or discontinuation when graded correctly.
Hassel JC et al. Eur J Cancer 2017.
Patients are penalised twice: by the skin toxicity itself and by the dose modifications that follow. Grading needs to be reproducible, photo-anchored, and fast enough to fit into a 7-minute oncology consultation. That is what Oncoderm is for.
What Oncoderm grades
Each sign has its own clinical anatomy: photographic in some cases, questionnaire-based in others. Oncoderm hides that complexity behind a single mobile-first interface, and produces a CTCAE-aligned grade in every case.
Skin redness, papulopustular rash, and acneiform eruption, the hallmark dermatologic toxicity of EGFR inhibitors.
Oncoderm’s erythema engine analyses tone, contrast and lesion density against a calibrated reference cohort. Output includes a heat-mapped overlay that lets the clinician verify the AI’s region of interest before signing the grade.
Pustular folliculitis and seborrheic-pattern dermatitis on the scalp; frequent but underreported on standard CTCAE checklists.
Three-angle capture catches lesions that a single dermoscopic shot misses. The model returns per-view confidence so the clinician can re-photograph an uncertain region rather than over- or under-grade.
Itch is the most disabling subjective symptom in cutaneous oncology toxicity, and it has no biomarker.
Four-question structured assessment combining the NRS-11 itch numeric scale with distribution, excoriation severity, and functional impact; collapses into a single CTCAE grade in real time on the device.
Stomatitis is the dose-limiting toxicity for many mTOR inhibitors, chemo-radiotherapy regimens and conditioning protocols.
Mucositis grading is binary in practice; patients either tolerate oral intake or they don’t. Oncoderm walks the clinician through a four-domain questionnaire and produces the CTCAE grade with a one-tap escalation flag for grades 3 – 4.
Inflammation of the periungual tissue, frequent with EGFR inhibitors and taxane regimens; under-detected because it tends to present late in the treatment cycle.
Paronychia grades on a three-factor questionnaire (visual finding, intervention level, ADL impact); the final grade is the highest of the three. The assessment is HCP-administered with the patient present, and no image AI is required.
And one more capability
Oncoderm also returns a ranked differential diagnosis with risk metrics from a single photograph, to support the clinician in deciding whether to refer or biopsy. This output is diagnostic decision support, not a CTCAE grade.
How it works
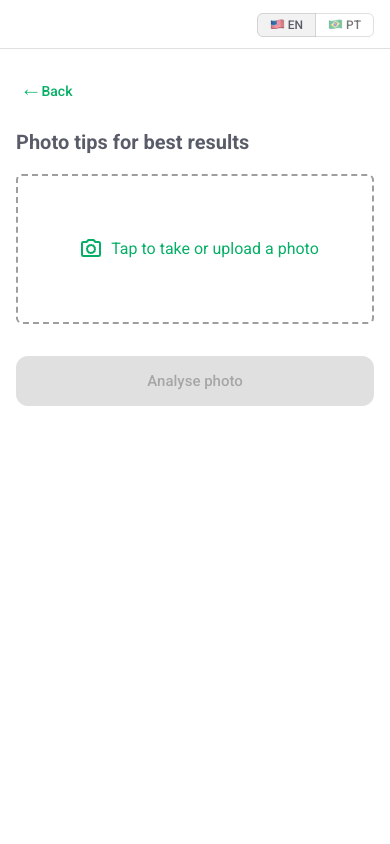
No login. No app store. A clinician opens the URL on any phone, tablet, or laptop and starts immediately.
For erythema and scalp rash, the camera UI guides standardised photographs. For pruritus and mucositis, four short structured questions.
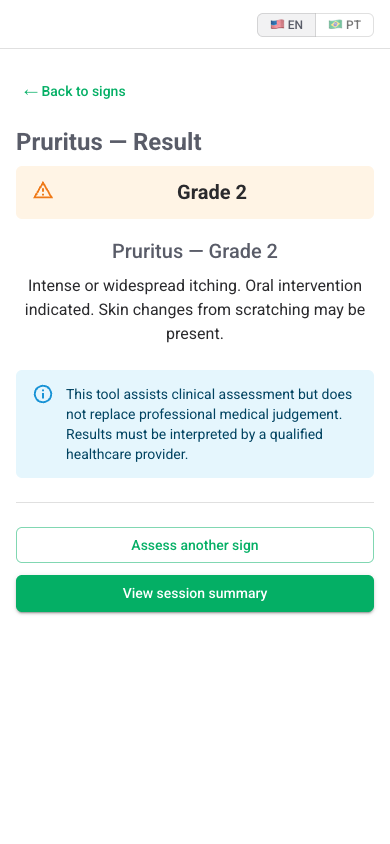
A CTCAE grade with rationale, anchored to the input. The clinician can override, attach a note, and export the assessment as a structured record.
Powered by Legit.Health+
Oncoderm calls into Legit.Health+, a dermatology AI platform with a portfolio of severity experts, regulatory documentation, and an eight-year track record in clinical settings. You get an oncology-tuned product on top of an infrastructure that already meets the bar.
Learn about Legit.HealthEarly access
We are onboarding a small cohort of oncology centres, dermatology services, and clinical-trial sites. Tell us about your setting and we will get back within two working days.